Pioneering Microbiology Testing Solutions
Your Trusted Partner in Ensuring Safety and Quality
At Sure-BioChem Labs, we understand the critical importance of accurate and reliable microbiology testing. Our state-of-the-art facilities and highly skilled team of experts are dedicated to providing top-notch microbiology testing services that cater to a broad spectrum of industries. Whether you're in pharmaceuticals, biopharmaceutical, food production, medical devices or cosmetics our services are tailored to meet your specific needs, ensuring safety, compliance, and quality at every step.
Industry-Leading Expertise
Our team comprises seasoned microbiologists, chemists, and quality assurance experts. They bring a wealth of experience and a passion for precision in every project they undertake. Their expertise is not just in conducting tests, but also in interpreting results and providing actionable insights.
Cutting-Edge Technology
We use the latest in microbiology testing equipment and methodologies. This commitment to technology means faster turnaround times, higher accuracy, and more detailed insights for your products and processes.
Customized Solutions
We understand that each client has unique needs. That's why we offer customized testing solutions designed to meet your specific requirements. Our team works closely with you to understand your challenges and objectives, ensuring that our services align perfectly with your goals.
Get a Microbiology Testing Quote
Fill out the Testing Quote Form and take the first step to
understanding the critical importance of accurate and reliable microbiology testing.
Microbiology Testing Quote Form
We will get back to you as soon as possible.
Please try again later.
Microbial Culture:
Refers to the method of multiplying microbial organisms by letting them reproduce in predetermined culture media under controlled laboratory conditions.
Pathogen Detection:
Involves identifying harmful microorganisms like bacteria, viruses, or fungi in samples from various sources.
Antimicrobial Susceptibility:
Tests to determine the effectiveness of antibiotics or antifungals against specific microbes.
Sterility Testing:
Ensures that pharmaceutical products or medical devices are free of microbial contamination.
Biochemical Tests:
Procedures used to identify bacteria species based on the biochemical activities of the organisms, like carbohydrate fermentation, enzyme production.
Molecular Diagnostics:
Techniques like PCR used for detecting sequences in DNA or RNA that might be associated with disease or pathogens.
Environmental Monitoring:
Testing for microbes in environments such as cleanrooms, food production and contract manufacturing organizations to ensure compliance.
Food Microbiology:
Testing to detect and control microbial contamination in food and beverages.
Water Testing:
Analysis of water samples for microbial contamination, including bacteria, heavy metal, volatile organic compounds (VOCs).
Bioburden Testing:
Assessment of the number of microorganisms on a product or a surface.
Endotoxin Testing:
Detecting the presence of endotoxins, particularly in pharmaceuticals and medical devices.
Clinical Microbiology:
Focuses on identifying and studying microorganisms that cause diseases in humans.
Ensuring Compliance and Safety
In today's regulatory landscape, compliance is key. Our microbiology testing services help you adhere to the strictest standards set by regulatory bodies such as the FDA, EPA, and USDA. We stay abreast of the latest regulations and standards, ensuring that your products not only meet but exceed the required safety and quality benchmarks.
Our Commitment to Excellence
We believe that excellence in microbiology testing comes from a blend of advanced technology, expert knowledge, and a deep commitment to our clients' success. That's why we've invested in cutting-edge equipment and continuous staff training, ensuring that every test we conduct is accurate, reliable, and timely.
A Partner in Your Success
At Sure-BioChem Labs, we see ourselves as more than just a service provider; we are your partners in success. Our team is committed to providing support and advice throughout the testing process, helping you navigate complex challenges and make informed decisions.
Industries We Serve
- Pharmaceuticals and Medical Devices: Ensuring the safety and efficacy of drugs, medical devices, and healthcare products.
- Food and Beverage: Guaranteeing the quality and safety of food and drink products.
- Cosmetics and Personal Care: Testing for harmful microorganisms in beauty and personal care products.
- Environmental and Water Testing: Analyzing water and environmental samples for microbial contamination.
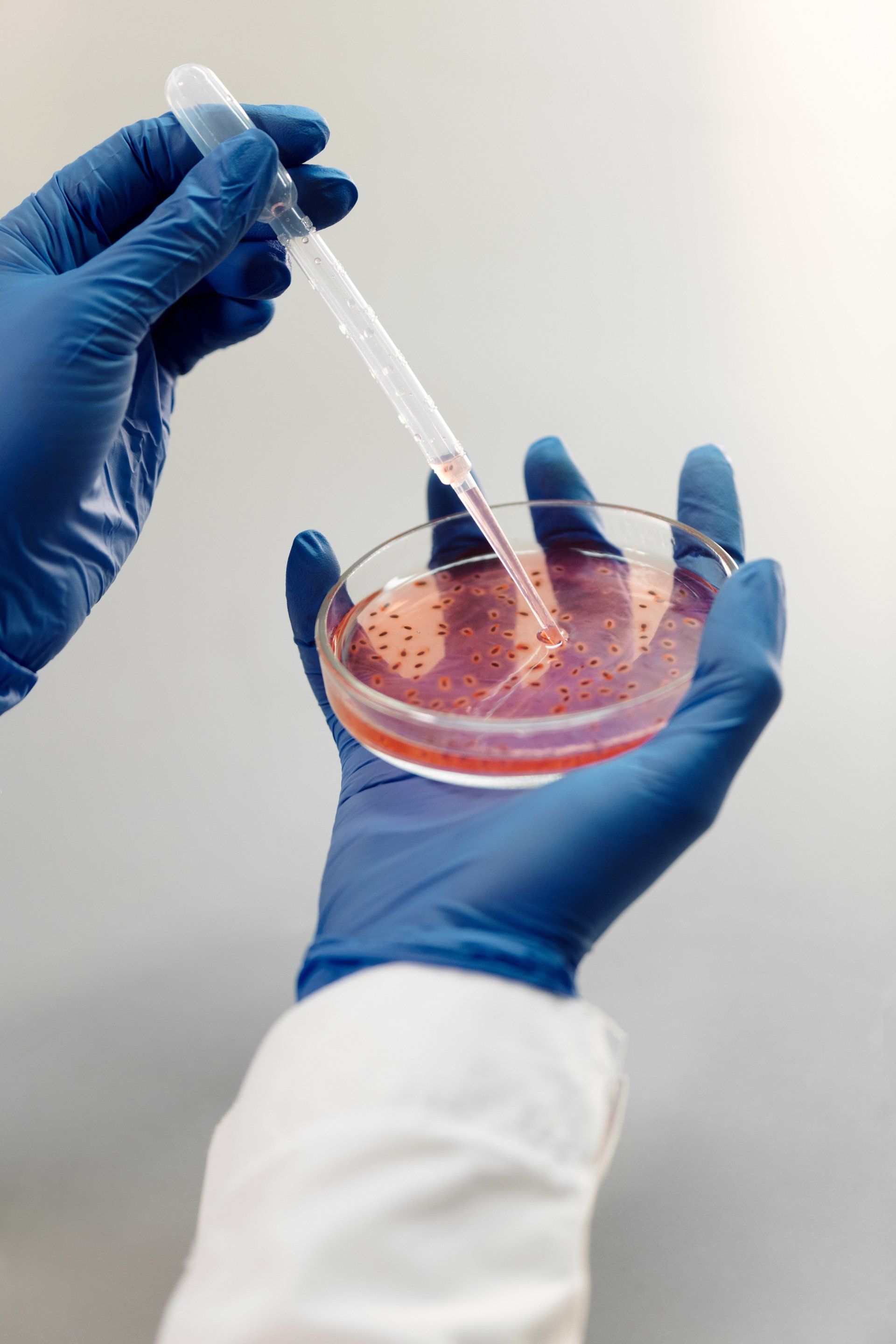


Our Promise to You
- Accuracy: Precision and accuracy are at the heart of everything we do.
- Timeliness: We understand the importance of timely results and strive to deliver on time, every time.
- Confidentiality: Your data and results are treated with the utmost confidentiality and integrity.
- Customer Support: Our team is always ready to assist you with any queries or concerns.
Get in Touch
Embark on a journey towards assured quality and safety with
Sure-BioChem Labs. Request a quote and learn more about how our microbiology testing services can benefit your business.
We're here to provide the expertise, technology, and support you need to thrive in today's competitive marketplace.
Interested in Working with
Sure-BioChem Laboratories
Follow Us
Sure-BioChem Laboratories offers top-notch analytical testing for various industries. Our advanced lab and expert team ensure reliable, quality results. We're committed to excellence, helping clients meet high standards in environmental, food, and pharmaceutical testing.
Headquarters:
1000 Atlantic Avenue
Camden, NJ 08104
PHONE: 888-398-7247
Main Menu









